Almost a century and a half ago, a visionary man described a potential role for Hydrogen:

As discussed in several recent articles, Hydrogen can be an attractive source of Clean Energy but it can also be an effective Energy Storage medium. Both the Upstream and Mining Industries have good applications for Hydrogen.
Sources of Hydrogen
Hydrogen is an important energy source for the Energy Transition since its combustion produces only water – but the production process needs to be considered. Popular terms for the method of production involve the colours Grey, Blue, Turquoise, and Green:
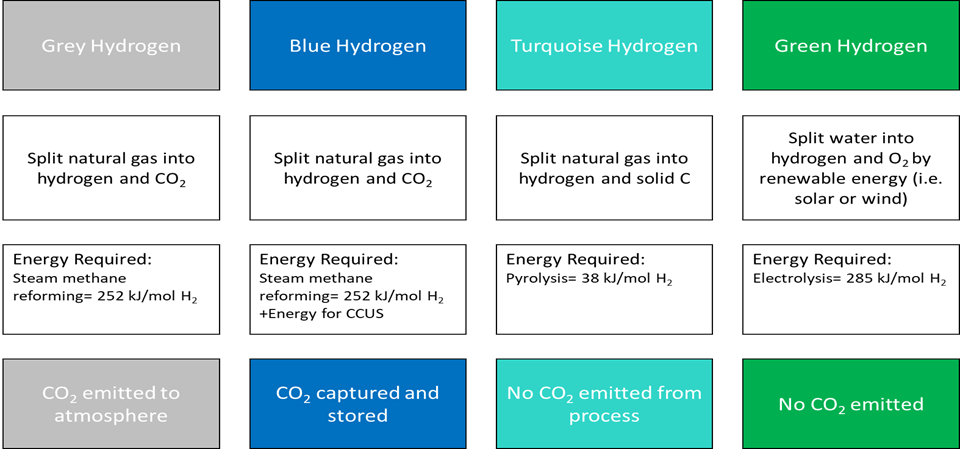
Hydrogen can be produced from natural gas in a process called “Grey Hydrogen” which involves thermal processes such as steam-methane reformation[1] or electrolysis powered by gas-fired power generation. These methods of production have been used commonly, but CO2 is produced in this process, so some improvements are necessary.
To be classified as “Blue Hydrogen” the CO2 needs to be captured, stored, and reinjected downhole (sequestered). Peter Coleman-Woodside CEO has stated “Blue Hydrogen is the key to building scale and lowering costs in Hydrogen transport and distribution, which will enable an earlier transition to renewable green Hydrogen, produced through electrolysis of water, powered by renewables. The earlier we can shift, the faster we can reduce emissions.”[2]
Another type of low-carbon Hydrogen produced from natural gas is called “Turquoise Hydrogen” and it involves pyrolysis which is a family of technologies being scaled up now. One such production route to Hydrogen is “catalytic thermal decomposition” where natural gas is heated up in a vertical reactor to high temperatures in order to generate Hydrogen whilst valuable nanostructured carbon black particles are simultaneously produced. Other pyrolysis production routes include “thermal and non-thermal plasma”, “thermal non-catalytic”, and “liquid metal” (passing methane through a bubble column reactor of molten liquid metal (>1000°C)). In all pyrolysis production routes there are no CO2 emissions associated with the processes. The carbon intensity of these process is dependent on the details of methane production and the source of energy for the reactors. But because energy potential is released from the four Hydrogen atoms (e.g. CH4) during processing, much less energy is required than splitting water (H2O) during electrolysis (since its Hydrogen atoms are already oxidized). This means pyrolysis is a very energy efficient means to produce Hydrogen.
“Green Hydrogen” is the production of Hydrogen through electrolysis powered by renewables power generation (i.e. Wind or Solar) – this is the most popular and environmentally compliant current manifestation of Hydrogen today. Some electrolysis technologies need purified water, but other technologies using seawater are advancing rapidly, as well as more efficient catalytic technologies for the electrodes.
Very early stage research is ongoing with Solar technology that splits water into Hydrogen and Oxygen (“Solar-Chemical Energy Conversion”) using semiconductor photocatalysts loaded with metallic cocatalysts (rod-shaped nanoparticles).[3] Very low energy conversion efficiencies for now, but progress is ongoing for the past few years to achieve eventual solar-driven photocatalytics splitting of water into Hydrogen and Oxygen.[4]
A sometimes overlooked potential source of Hydrogen is the by-products of hydrocarbon fractionation in refineries.[5] Refineries produce and separate Hydrogen for their own processes, but Hydrogen for export appears feasible also. Effluent and purge streams from refinery hydrocrackers and hydrotreaters have Hydrogen.[6] It has been estimated that significant amounts of this lost Hydrogen could be recovered with better use of membranes. Companies like Air Products, Air Liquide, and Honeywell have this technology which operators can use. The membrane technology was identified back in 1993.[7]
Hydrogen Production
For purposes of this article, we can assume one of two Green methods are used to produce Hydrogen using Renewables produced electricity:
- PEM Electrolysis;
- Reversible Solid Oxide Fuel Cell (RSOFC)(High-Temperature Electrolysis (HTE) steam system).
1. Electrolysis is a well documented method using electricity to breakdown water into Hydrogen and oxygen. Proton Exchange Membrane (PEM) technology is a good choice for the electrolysis system. Typical PEM layout as shown[8] with a photograph of Siemens Silyzer 300 unit for PEM electrolysis:
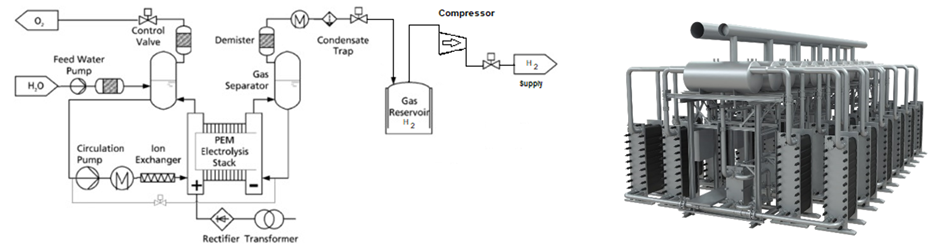
Siemens Silyzer 300 PEM units were installed in the Voestalpine-Linz, Austria Steel Plant where Hydrogen is produced as process gas for steel production. The 6 MW facility (12 stacks x 50 cells/stack) produces 1200 m3/hr of Hydrogen with a nominal efficiency of ~75%. Previously Siemens had installed Silyzer 200 PEM units at the Mainz Energy Park-Germany (right photo) to produce hydrogen for mobility and industry including grid support.[9] The 3.75 MW nominal /6 MW maximum unit (3 stacks @ 2 MW max /stack) produced 675 m3/hr of hydrogen with a nominal efficiency of 65-70%. Hydrogen outlet pressure was ~35 bar. Future large scale applications of the Silyzer 300 units are Module Arrays with 24 modules rated at 17.5 MW, system efficiency ~75%, producing 340 kg/hr of Hydrogen (0.02kg/kWh).[10]
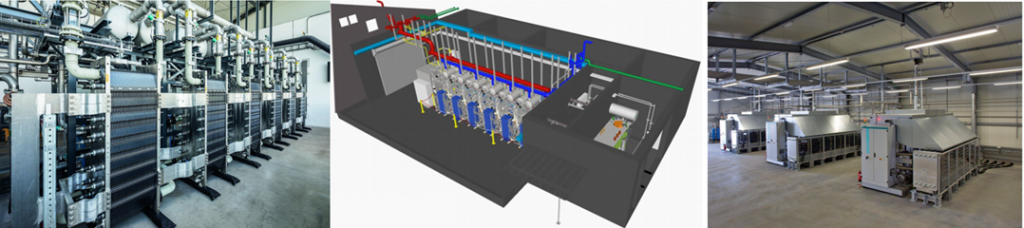
ITM Power makes a series of modular solutions for PEM electrolysers[11] ranging from (1) HGas1SP=~0.7MW to produce ~270 kg/day; (2) HGas2SP=~1.3MW to produce ~540 kg/day; (3) HGas3SP=~2.0MW to produce ~800 kg/day; to (4) HGasXMW modular units (3-5 stacks @ ~2 MW/module) (so 6 MW facility produces 2400 kg/day or 100 kg/hr).

A 10 MW ITM electrolyser facility was installed at the Shell Wesseling refinery site within the Rheinland Refinery Complex in Germany[12]:
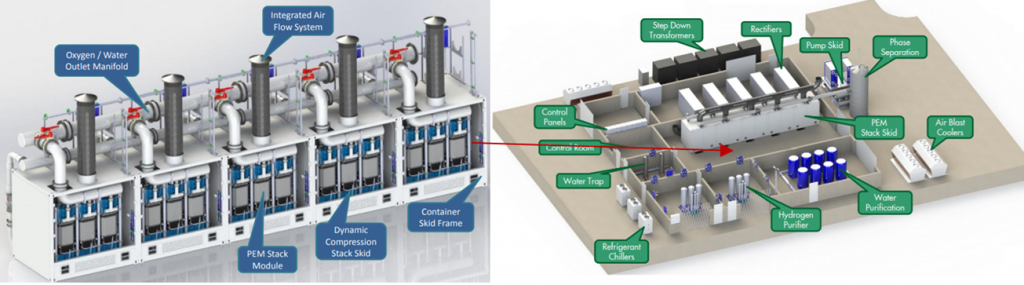
Project cost was ~€20 million including integration into the refinery to produce ~1300 tonnes/year Hydrogen. The 10 MW stack skid comprised 5 x 2 MW submodules. Stack efficiency was ~ 45-55 kWh/kg (~0.02 kg/kWh).[13]
2. RSOFC is an interesting method to use solid oxide fuel cells able to work in two directions: initially one direction to produce Hydrogen and subsequently the reverse direction to produce energy from the Hydrogen. The solid oxide fuel cells (SOFC) act as a High-Temperature Steam Electrolyser.
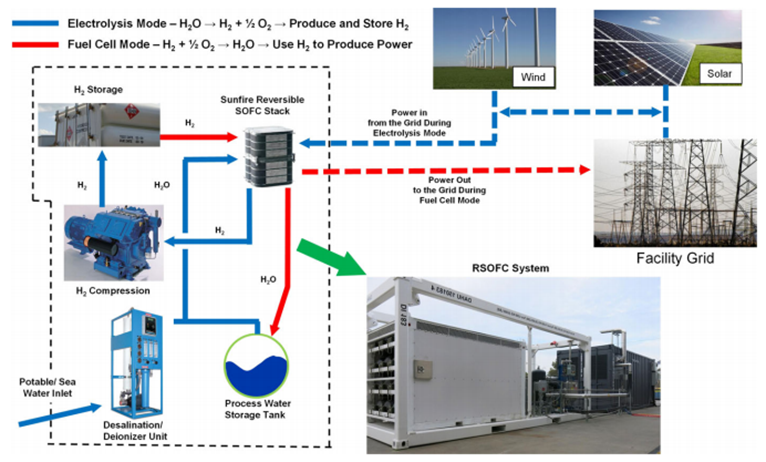
Boeing and Sunfire-HyLink developed the scalable, modular solution shown above.[14] It was sized at 2 x 100kW to produce ~50Nm3/h Hydrogen output. Reversing the operation produced 2 x 20 kW electricity with a roundtrip efficiency ~45%. The amount of storage is able to be configured as desired to have energy storage duration as long as desired. Sunfire also worked with Salzgitter to install a more powerful reversible high-temperature electrolyser for their steel plant – 150 kW electrolyser power input to produce ~40 Nm3/h Hydrogen at electrolyser efficiency >80%. Phase 2 called GrInHy2.0 has recently been installed with 720 kW power to increase Hydrogen production up to ~200 Nm3/h (18 kg/h).[15] Hydrogen is produced at under 7€/kg with electrolyser CAPEX < 4,500 €/(kgH2/d).
Hydrogen Storage
Hydrogen once produced is able to be stored to be used to produce energy later when needed. There are physical and material-based mechanisms to store the Hydrogen[16]:
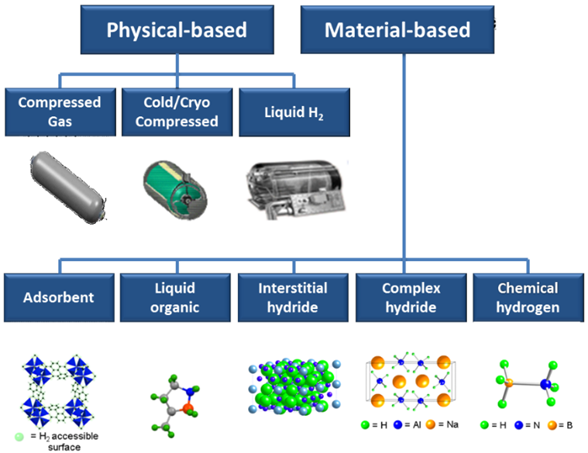
Two good methods used for hybrid microgrid Hydrogen storage are (1) compressed gas and (2) interstitial metal hydrides. Other methods like cold/cryogenic compressed storage or liquified nitrogen are more CAPEX and OPEX intensive and therefore may not be appropriate for remote microgrid applications.
For the compressed gas method of storage, a wide range of storage pressures are possible, associated with the type of storage tanks and type of withdrawal (i.e. use in fuel cell, use in generator, or injection into natural gas pipeline).

Higher pressure (~300-500+ bar) storage methods and tanks are also able to be used with additional compression facilities. A different risk profile and this storage solution may not needed except for transportation refuelling:

For the interstitial metal hydride method of storage, produced Hydrogen is introduced into metal storage containers where compacted powdered elemental hydrides (consisting of various compounds such as lanthanum, nickel, aluminium, boron (borohydrides), and/or magnesium hydride[17]) were packed ready to store Hydrogen through adsorption.[18] Subsequent Hydrogen desorption requires heat input which can often be recovered from the production process. A major benefit of this storage method is its efficient storage of Hydrogen at relatively low pressures and volumes. Two equal sized containers can contain the same volume of Hydrogen – high pressure (182 bar) compressed Hydrogen in one and low pressure (10-20 bar) adsorbed Hydrogen in the other. Hydrogen is released from storage through desorption using thermal heat (45-65°C) with outlet pressure down to <5 bar. Storage capacity of this kind of system is 1.5 kg (~50 kWh) Hydrogen per 100 kg of the metal hydride compound material.[19]
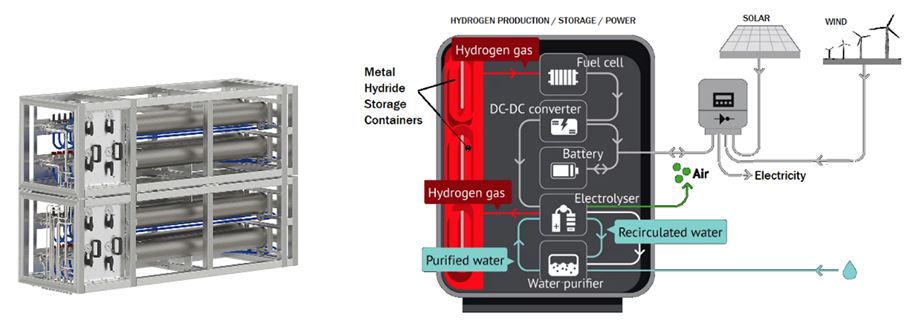
Energy Production
Hydrogen has been produced with electricity from Renewables and it has been used as a storage medium. Now when needed by demand unable to be satisfied from intermittent Renewables, it needs to be used to produce energy. Several ways are possible or under development:

Hydrogen Fuel Cells can be assumed for use in typical Upstream and Mining applications until such time as other technologies become more commonly deployed. The assumed facility could have a modular series of stacks for Hydrogen production, storage, and energy production. With Renewables producing the initial energy, these Hydrogen solutions would ensure the complete facility would be 100% RE during intermittencies.
Technical Challenges on Plant Equipment
Technical challenges remain for the risk to some steels of Hydrogen-Induced Stress Corrosion Cracking (HISCC). Under certain conditions Hydrogen can degrade the fracture behaviour of many structural alloys, including many stainless steels, by causing brittle failure to occur caused by interface separation of grain boundaries. The use of HISSC resistant steel as a construction material would be the preferred way to reduce this risk, but some equipment and piping may not be suitable. Some blending of Hydrogen has been used (5-15%) into natural gas without substantial negative impact on valves or equipment infrastructure. The latest European recommendation is <6% compositional blending now, with targets on infrastructure materials to rise to <10% by 2030 and <30% eventually. Some technical advice on the use of conventional steel piping has recommended limiting operating pressure stresses to <30 SMYS or <20% SMUTS for blended natural gas + Hydrogen service.
Emissions
The energy density of blended natural gas + Hydrogen may need to be considered in calculating CO2 emission “savings” if this fuel was used for power generation instead of pure Hydrogen:
- 1 cubic metre of natural gas will provide 35.8 million Joules of energy;
- 1 cubic metre of Hydrogen will provide 10.8 million Joules of energy;
- A 70%/30% blend will therefore have 28.3 million Joules of energy so 1.26 cubic metres of blended gas will have to be combusted to get the same energy release as pure natural gas;
- So natural gas combusted will be 1.26 x 0.7 = 0.88 cubic meters which is a 12% reduction in CO2 not 30%.
Closing
The Upstream and Mining Industries have significant power demands in running their facilities. The Energy Transition challenges us to utilise Clean Energy sources. Renewables (Wind and Solar) offer such a source of Clean Energy, but they can be intermittent. In order to maintain grid stability and avoid curtailments, we need to have large capacity, long-duration Energy Storage Systems. Hydrogen offers a potential route to achieve the goal of 100% RE.
[1] https://www.energy.gov/eere/fuelcells/Hydrogen-production-natural-gas-reforming
[2] https://www.theguardian.com/environment/2019/apr/08/the-perfect-storm-woodside-energy-and-siemens-invest-in-australias-Hydrogen-economy
[3] https://spectrum-ieee-org.cdn.ampproject.org/c/s/spectrum.ieee.org/energywise/energy/renewables/solar-closing-in-on-practical-Hydrogen-production.amp.html
[4] https://pubs.rsc.org/en/content/articlehtml/2015/ta/c5ta05784a
[5]http://kchbi.chtf.stuba.sk/upload_new/file/Miro/Proc%20problemy%20odovzdane%20zadania/Galis%20a%20Witosov%C3%A1/Hydrogen%20management%20in%20refineries.pdf
[6] https://www.researchgate.net/publication/46027587_Hydrogen_Recovery_from_Refinery_Off-gases/link/02e7e53ba7f9290b29000000/download
[7] https://www.osti.gov/servlets/purl/10182536
[8] https://www.sciencedirect.com/science/article/abs/pii/S136403211731242X
[9] http://www.scandinavianhydrogen.org/wp-content/uploads/2016/11/2_Manfred-Waidhas.pdf
[10] https://australia.energyandmines.com/files/Case-Study-Opportunities-and-Challenges-of-Integrating-Hydrogen-into-a-Mining-Hybrid-Warner-Priest-Siemens.pdf
[11] https://www.itm-power.com/hgas1se ; https://www.itm-power.com/hgas2se ; https://www.itm-power.com/hgas3se ; and https://www.itm-power.com/images/Products/HGasXMW.pdf
[12] https://www.itm-power.com/item/39-10mw-refinery-hydrogen-project-with-shell
[13] https://www.eu-japan.eu/sites/default/files/imce/shell_presentation_at_eu-_japan_energy_seminar_april_2019_1.pdf
[14] https://onlinelibrary.wiley.com/doi/full/10.1002/fuce.201600185
[15] https://www.green-industrial-hydrogen.com/project/grinhy-project and https://www.green-industrial-hydrogen.com/
[16] https://www.energy.gov/eere/fuelcells/Hydrogen-storage
[17] https://www.sigmaaldrich.com/materials-science/alternative-energy-materials/Hydrogen-storage-alloys.html
[18] https://www.sciencedirect.com/science/article/pii/S0360319919302368
[19] https://www.gknpm.com/en/innovation/hydrogen-technology/hy2green/
[20] https://www.clarke-energy.com/2019/hydrogen-future-fuel/
[21] https://www.wartsila.com/media/news/05-05-2020-wartsila-gas-engines-to-burn-100-hydrogen-2700995
[22]https://www.man-es.com/docs/default-source/press-releases-new/20200625_man-es_pr_cr-report_2020_en.pdf
[23] https://www.powermag.com/high-volume-hydrogen-gas-turbines-take-shape/