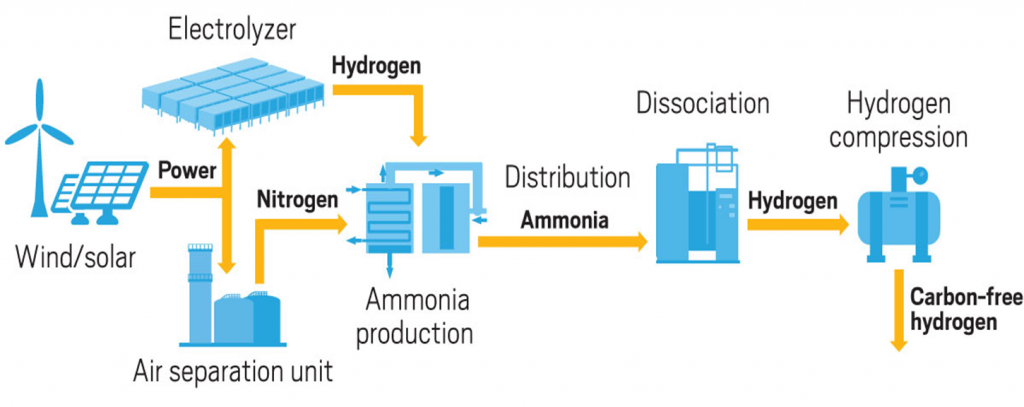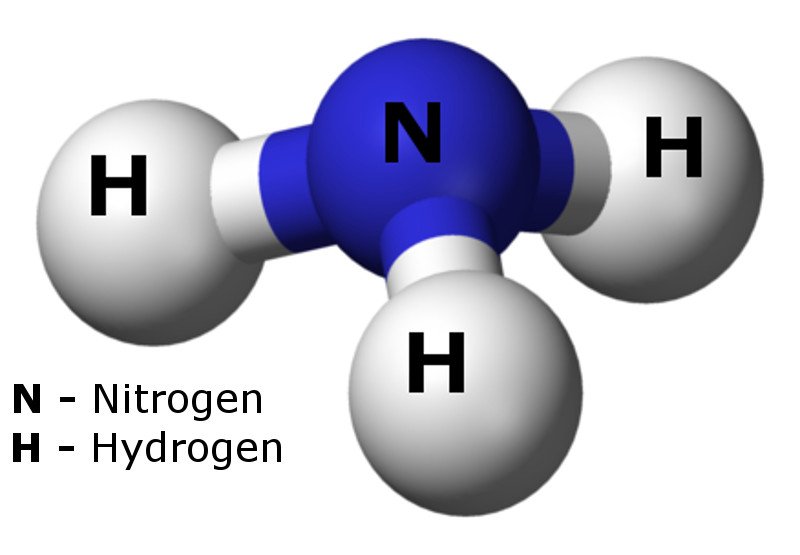
Ammonia (one nitrogen atom bonded to three hydrogen atoms) has an energy density almost twice that of liquid hydrogen and it is easier to ship and distribute.
Conventional ammonia production using the Haber-Bosch method is very energy intensive and produces significant carbon dioxide emissions.
One production process that uses renewable energy powered electrolysers to produce hydrogen and oxygen. Then the hydrogen is processed in a conventional Haber-Bosch method. This reduces the carbon footprint by 50%, but further improvement is being pursued.
A reverse fuel cell concept uses renewable energy to drive a chemical reaction that produces green ammonia. Water reacts at the anode to make hydrogen ions (H+), which migrate to the cathode where they react with nitrogen (N2) to form ammonia. The reaction is efficient, but being developed to be faster with ionic liquid electrolytes.